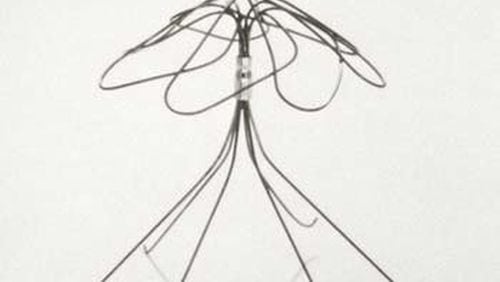
A Savannah woman whose medical implant fractured and moved, leaving a broken wire permanently stuck in the blood vessel between her heart and lungs, has lost her case against the company that made the device.
More than 3,000 patients have sued the device maker, Bard, over a metal filter implant. The filter is placed in a big vein leading to the heart in order to catch blood clots, but the patients said the device broke inside them or shifted position. The Savannah patient, Doris Jones, was among five whose lawsuits were chosen to be tried as "bellwether" cases for the group, and the first to lose.
Her lawyer, Lincoln Combs, said she would appeal to the 9th U.S. Circuit Court of Appeals, one step below the U.S. Supreme Court.
“We’re extremely disappointed in the result, but respect the jury’s decision,” Combs said in an email. “Unfortunately, we believe the Court made mistakes in what evidence we allowed the jury to hear, and believe if he had ruled differently the outcome would have been different as well.”
Jones’ is the second bellwether case; the first involved another Georgia patient, Sherr-Una Booker. Booker won her case this spring when a jury deliberating for less than six hours awarded her $3.6 million from Bard and $400,000 from a doctor.
“We won’t speculate on why different juries came to different conclusions,” Troy Kirkpatrick, a spokesman for Bard and its new owner, Becton Dickinson, wrote in an email. “I can tell you that we believe that this jury came to the right conclusion by recognizing that any implantable medical device carries inherent risks, but also provides clinical benefits that outweigh those risks.”
In Jones’ case, the judge quashed some evidence about deaths under early versions of the filter.
The federal Food and Drug Administration did not require Bard’s filter to be tested in humans to make sure it was safe before it went to market, and so the company didn’t.
The accusers suggest that they ended up being the test subjects. They allege that Bard introduced its filter to the market quickly in a race to gain profits. After people started dying following malfunctions, they say, the company quietly redesigned the filter, never doing a public recall or adequately warning the public. Bard introduced six versions of the filter over the space of a decade.
Bard counters that those redesigns were the natural evolution of the product, and that the government never suggested a recall. They say all companies’ filters have risks but they save thousands of lives when people who have blood clots can’t take blood thinners.
The judge overseeing the mass of Bard filter cases in federal court in Arizona, U.S. District Judge David Campbell, ruled that when it came to evidence about the earliest filter version, the Jones jury could hear about malfunctions but not about the deaths related to certain malfunctions.
Campbell found that the deaths had “marginal” relevance to Jones’ argument. He gave several reasons for his ruling, including that that type of malfunction was not known to cause deaths by the time Jones’ redesign came around, and the deaths that did occur were five years before she got hers. Booker’s jury was allowed to hear about the deaths.
In all the cases being heard in Arizona, Campbell ruled that the juries cannot hear about a jarring report commissioned by Bard’s lawyers in reaction to those first malfunctions. The company’s internal report disclosed that multiple deaths had occurred related to the filter, and it suggested that the Bard filter was more dangerous than most other filters on the market and that more study was “urgently warranted.”
Campbell’s reasons included that the report, commissioned by the company’s lawyers rather than its medical staff, was subject to attorney-client privilege.
All along, the company has argued that it complied with the FDA's rules and more. The device was legally able to skip the most rigorous vetting, undergone by devices that receive "approval." Instead, the filter was allowed to receive "clearance" — a process meaning that it did not have to be tested in humans first to demonstrate it was safe. It just had to demonstrate it was similar to a device already on the market and undergo other types of tests. The filter was tested in humans to make sure it could safely be retrieved, but not to make sure it was safe after implantation.
Company representatives say that later versions of the filter have since been tested on humans for safety. In addition, the company “provided appropriate disclosure of (the) risks and benefits to Mrs. Jones’ physician,” Kirkpatrick added.